The focus of our group is to define which cellular and molecular niche factors regulate hematopoiesis under various insults, such as inflammation and hematopoietic malignancies. We use cell imaging, single-cell transcriptomics, and genomics to chart the molecular blueprints of the bone marrow niche and define their precise contribution to hematopoietic stress responses in mouse and man.
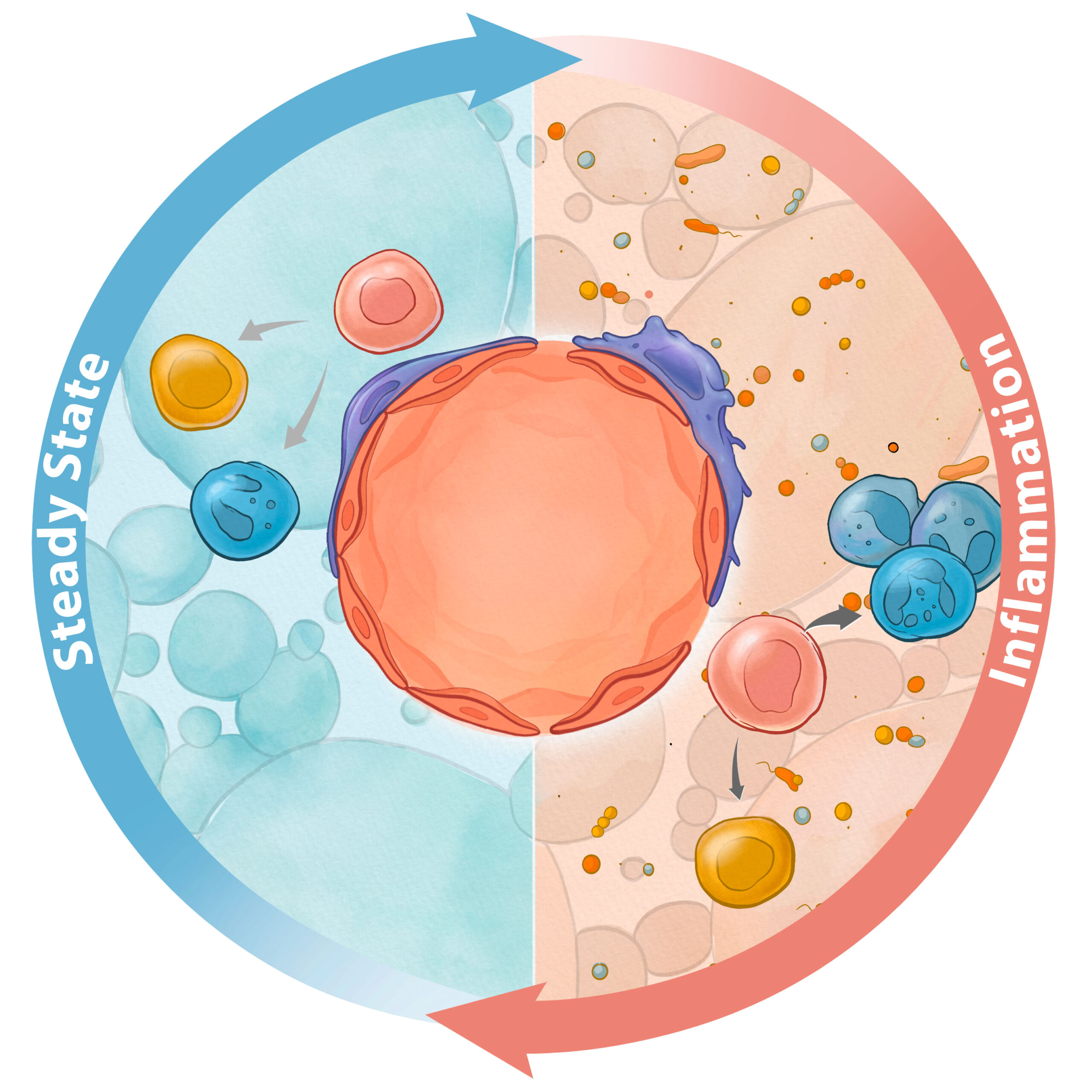
Stress-mediated Bone Marrow Niche remodeling
The hematopoietic system can rapidly and precisely adapt to varying perturbations such as inflammation and cytopenia. The bone marrow niche components sense inflammatory stimuli and reshape hematopoietic output accordingly. How does molecular dialog between hematopoietic cells and the bone marrow niche change in the context of stress perturbations? How do niche subsets coordinate the resolution of inflammation in the bone marrow? How does continuous stress contribute to the bone marrow niche dysfunction, and are those injuries reversible? We aim to define the impact of stress on bone marrow niche biology, ultimately leveraging this knowledge to develop novel therapeutic interventions to improve hematopoiesis.
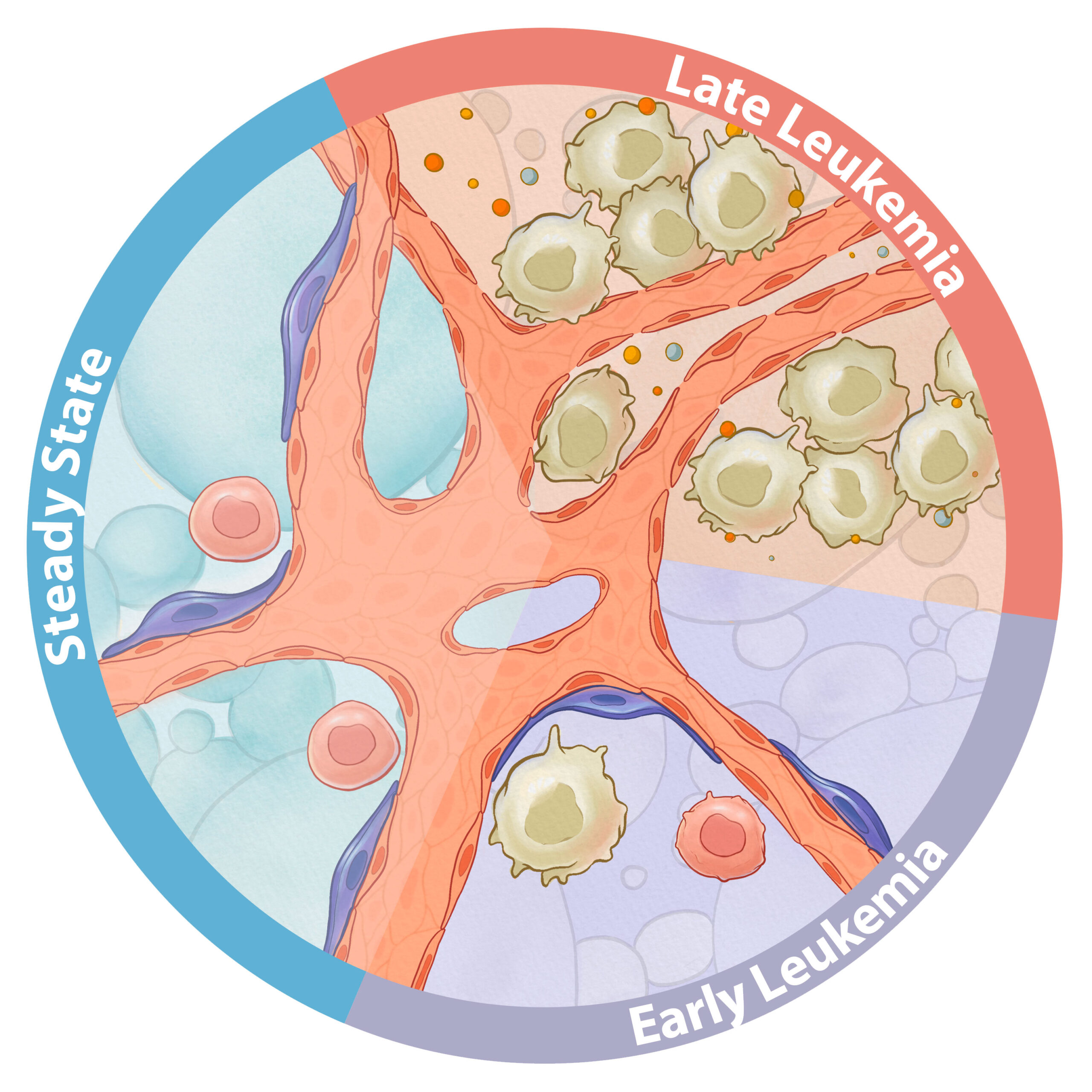
Leukemic Niche
Large-scale sequencing efforts aimed at elucidating the genetic makeup of T cells acute lymphoblastic leukemia at disease diagnosis and relapse have not been able to inform targeted treatment strategies or predict relapse. Accumulating evidence suggests that similar to normal hematopoietic stem/progenitor cells , leukemic cells receive key growth and survival signals from the bone marrow niche. We do not fully understand which bone marrow microenvironment signals and cellular fractions facilitate leukemia progression and resistance to chemotherapy. How do leukemic cells rewire the bone marrow microenvironment? Which factors produced by the niche are required for tumor growth and maintenance? Can we target niche-leukemia interactions? We believe that this work will uncover novel subpopulations and cellular interactions that can be targeted to enhance treatment responsiveness and prevent leukemic relapse.
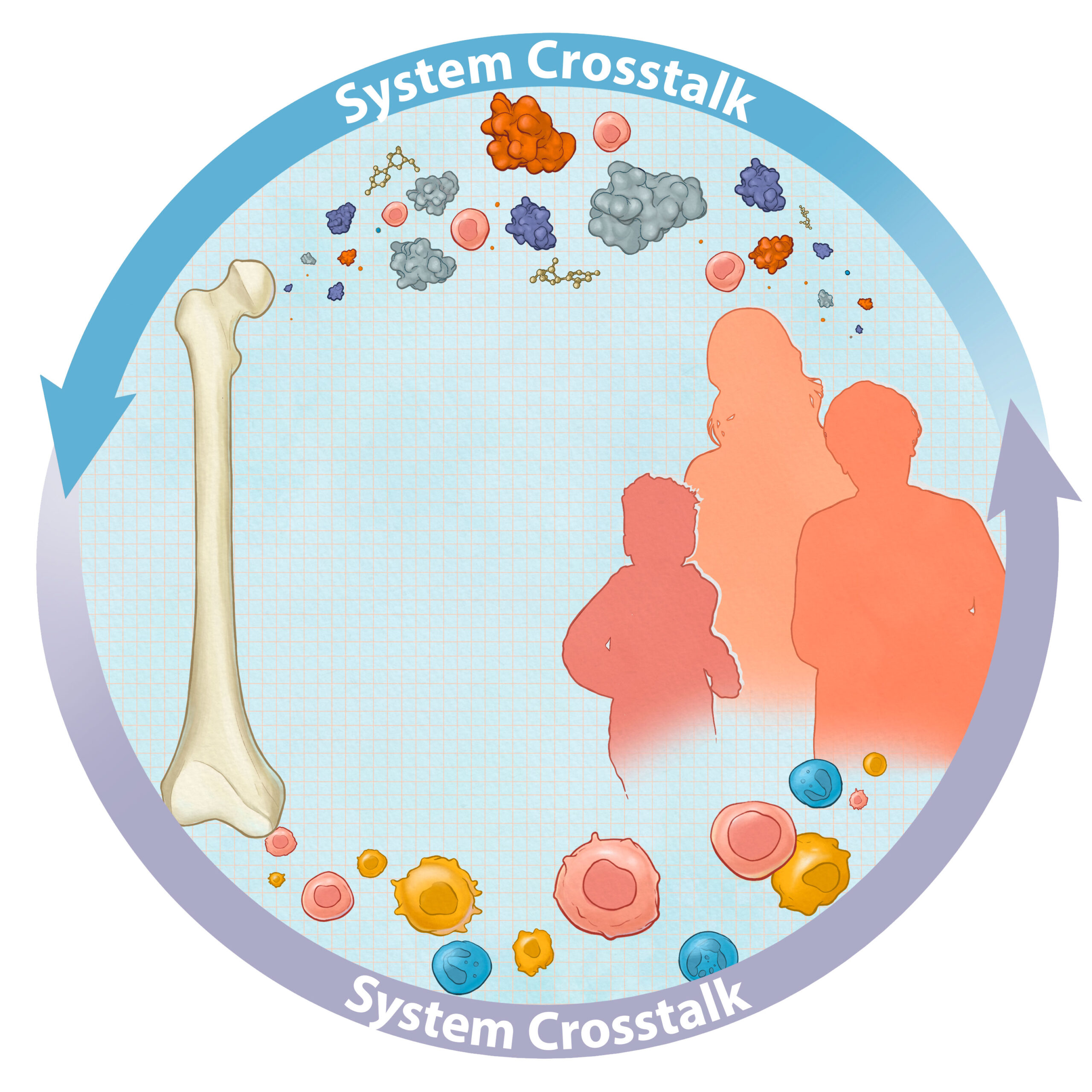
In and Out of the Bone
In addition to hematopoietic malignancies, many other tumor types including prostate, breast and lung metastasize to and seek refuge from chemotherapy in the bone. Despite considerable advances in therapies targeting these cancers, once tumor cells have metastasized to the bone, they are generally untreatable. Our goal is to define molecular mechanisms driving bone marrow involvement in malignant processes. Understanding the cell-cell interactions that facilitate bone marrow colonization is an urgent clinical need and will allow for the targeted design of improved therapeutic strategies.
Hematopoietic cells are directly involved in functional repair in response to injury in retina, brain, spinal cord, and infarcted myocardium. Understanding the cellular and molecular factors that orchestrate hematopoietic response to radiation injury is critically important for developing strategies that promote tissue repair. Together with our colleagues at Princess Margaret Cancer Center, we aim to delineate how hematopoietic cells sense damage in distal sites of the body and modulate regeneration.